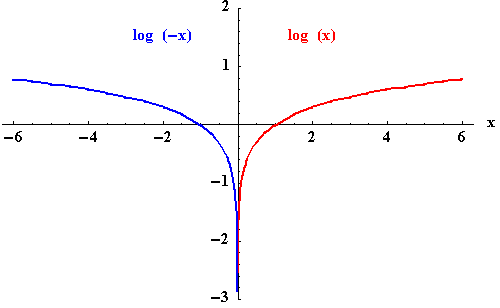

If b=0 then f(x)=0 (a horizontal line) and g(x) is not a function because it is a vertical line. On the whole, log amps do not behave in this way. We can have just one divide: Example: What is log8(0.125). Note: log_c(b) = log_c(1/c) = log_c(c -1) = -log_c(c) = -1 A negative logarithm means how many times to divide by the number. Example: Calculate the value of each of the following: a) 1og2 64 b) log9 3 c) log4 1 d) log6 6 e) log8 0. G(x) = log_b(x) = log_c(x)/log_c(b) = -log_c(x) where the graph of the 01 case about the x-axis. The base of logarithms cannot be negative or 1. If b=1 then f(x)=1 (a horizontal line) and g(x) is not a function because it is a vertical line.į(x) = b x = (1/c) x = 1/c x = c -x where the graph of the 01 case about the y-axis The natural exponential function is given as: e x. where the natural log or ln is the inverse of e.
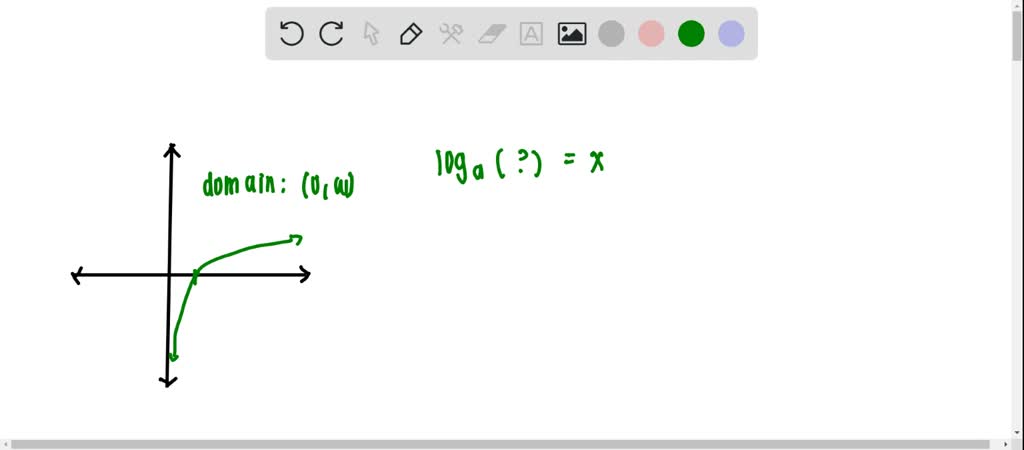
Mathematically, the natural log of a number x is written as: log e x ln x. This equation yields two negative logarithms therefore 2 cannot be a a. A natural logarithm is a special form of logarithms in which the base is mathematical constant e, where e is an irrational number and equal to 2.7182818. WolframAlpha shows the graph of the logarithm g(x) b=e with real and imaginary projections and for several values of b. The natural logarithmic function y log e x is abbreviated y ln x and is the. WolframAlpha shows the graph of the exponential f(x) for several values of b note as x approaches -infinity f(x) drops toward zero from the positive side faster than any other type of function and as x approaches +infinity it grows faster than any other type of function. Note see Wikipeida for complex exponential and logarithm function definitions] Note logarithm base conversion: log_c(x) = log_b(x)/log_b(c) The real inverse is the logarithm function g(x)=log_b(x) where b is the base, domain = (0,+inf), range = (-inf,+inf). To measure earthquake intensity on the Richter scale.
#Negative logs free
The real exponential function is f(x)=b x where b is the base, domain = (-inf,+inf), range = (0,+inf). The pH is the negative logarithm of the concentration of free hydrogen ions. You are clearly interested in negative bases. I don't know if you will find this useful or interesting but here is a summary of what others have said and some WolframAlpha pages:
